Expressing Concentrations Tutorial
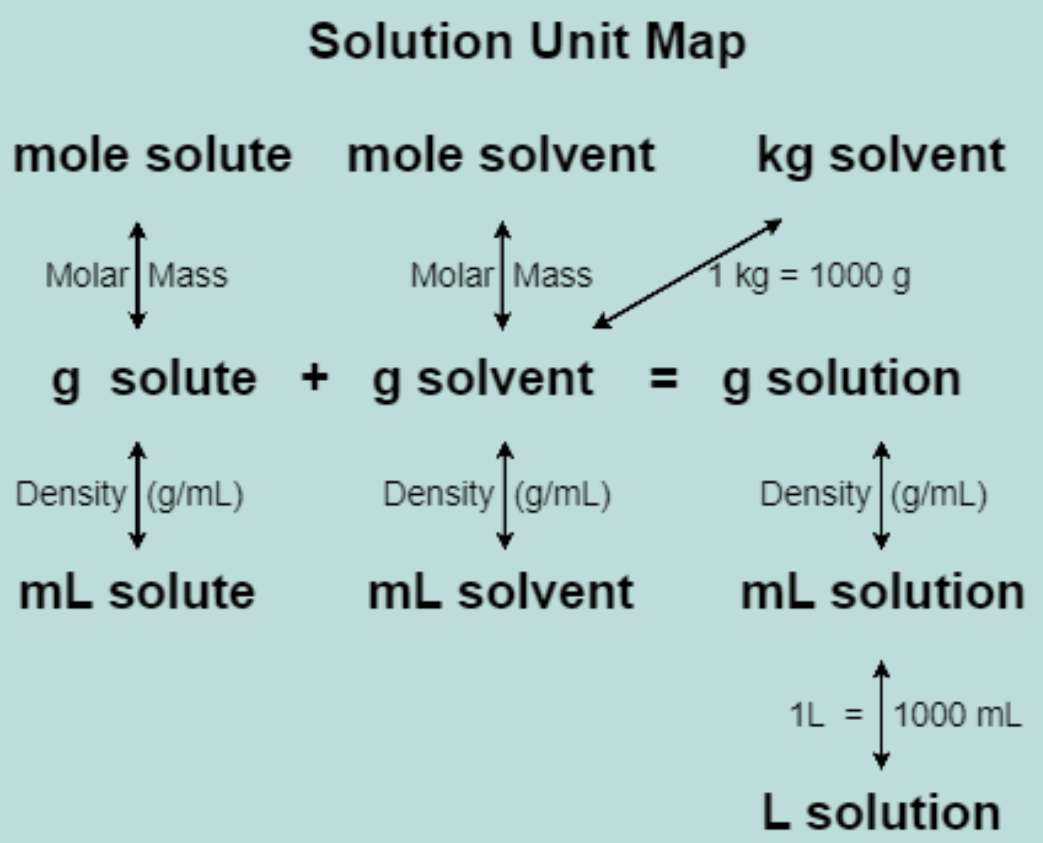 This "map" shows the connections between units used in concentation calculations.
This "map" shows the connections between units used in concentation calculations.
The Expressing Concentrations Tutorial is an interactive animation that defines solutions (solute + solvent), guides students in the creation of a Unit Map (shown), and presents several types of calculations that convert one concentration unit into another concentration unit. Chem21 animations provide a safe environment for learning . . . . students receive full credit when they correctly complete the animation - even with multiple restarts. This learning engagement is typically a lecture assignment, but can be assigned as a post-lab activity for labs like Oscillating Chemical Reaction, Molarity of Vinegar, etc.
Assigning the same tutorial in both lecture and lab is a great way to connect learning between the two courses.
In this tutorial . . . .
- students learn that solutions are comprised of a solute and a solvent.
- the Law of Conservation of Matter (CoM) provides the central relationship . . . .
g Solute + g Solvent = g Solution
- Th CoM equation is "expanded" into the Soution Unit Map shown to the right.
- Students re-create this map by dragging / dropping units in the correct spot.
- The animation ends with a series of solution calculations where students use their map to express a given concentration in Molarity, molality, mass percent, ppm, mole fraction, or Normality.
- If the student enters an incorrect answer, they are shown the step-by-step solution, however, they must restart the animation.
- Students receive full credit when they correctly complete the animation - even with multiple restarts.

