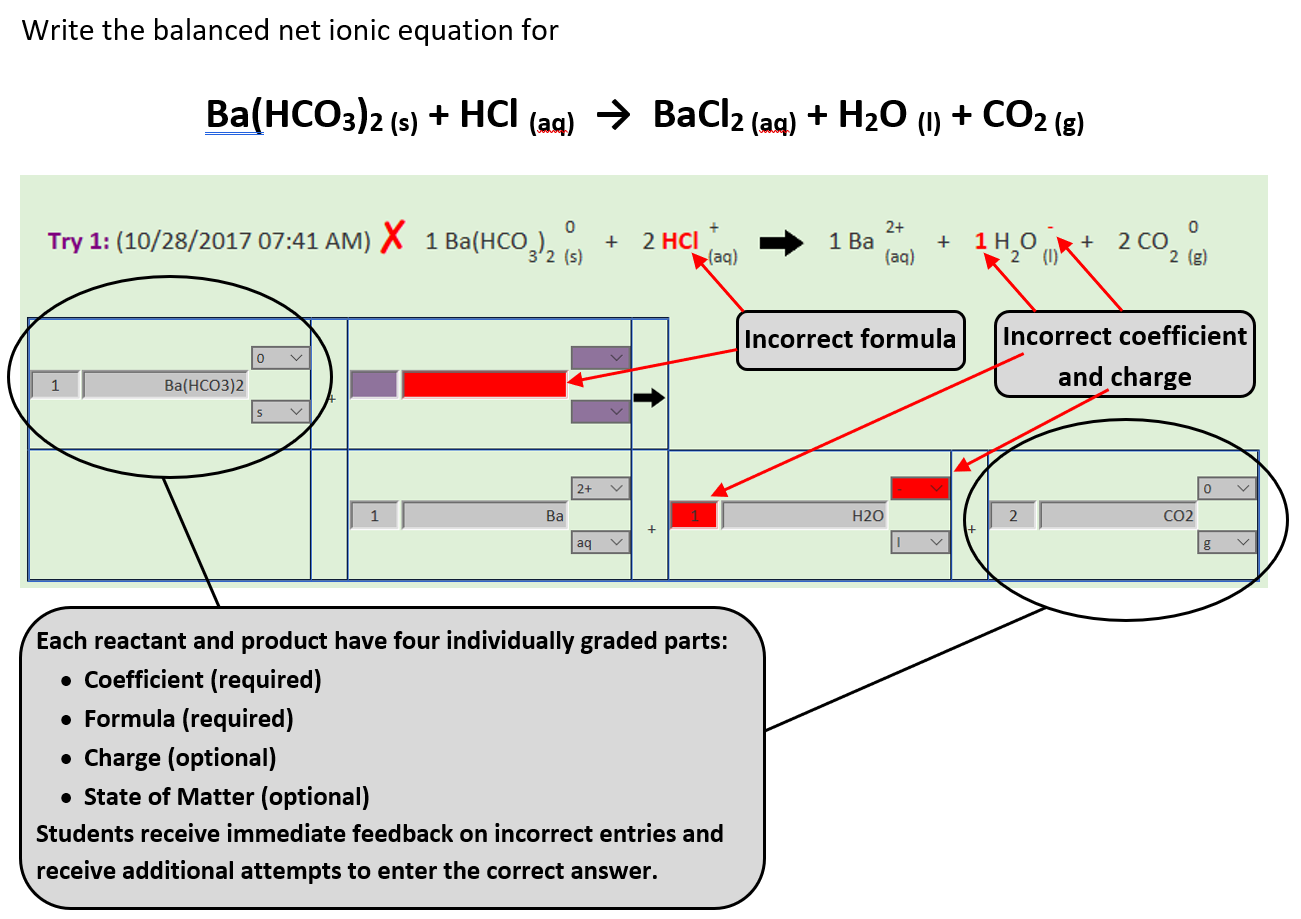
Balancing Chemical Equations
Writing and balancing molecular, ionic, and net ionic equations is a fundamental chemistry skill, and immediate feedback is key to mastering it. Each reactant and product includes four parts: a required coefficient and formula, plus optional charge and state of matter.
As students enter these components using text boxes and dropdowns, the program instantly assembles a properly formatted equation, displaying subscripts for formulas and states, and superscripts for charges.
A More Information link provides guidance on balancing these equations, expanding a collapsible section to save space; it can be hidden again using the Hide Information link.
Directions For Writing Molecular, Ionic, And Net Ionic Equations:
- Molecular Equation: predict products and balance coefficients so all atoms are conserved (Law of Conservation of Matter).
- Ionic Equation: split strong acids, strong bases, and soluble salts labeled (aq) into ions. Multiply coefficients by subscripts (e.g. 2 Na2SO4 → 4 Na+ and 2 SO42-). Do not split substances labeled (s), (l), or (g), or weak acids and bases.
- Net Ionic Equation: cancel spectator ions (unchanged on both sides) and simplify coefficients to the lowest whole-number ratio.
- Reminder: include charges and states for all species; neutral molecules have a charge of zero.