Homework
In modern classrooms, critical thinking is typically assessed through homework, quizzes, and exams. In-class work has the advantage of supervision, ensuring that submissions reflect the student's true ability. However, online homework can also provide a reliable assessment when designed properly.
- Problem Groups - a problem group contains about 10 similar questions, from which several are randomly assigned to each student. For example, if homework is drawn from 10 problem groups with two problems per group, each student receives a 20-question assignment, and any two students will share only about 20% of the same questions. While this once created sufficiently unique assignments, the widespread availability of solution websites and free AI tools can reduce homework to simply obtaining the correct answer rather than engaging in the critical thinking needed to solve the problem.
- The Coach Approach - introduces AI-Proof learning. Each student has the same homework problem set and they are encouraged to work together with classmates, tutors, instructors, internet resources, and even AI. However, the homework assignment has some significant changes:
- homework is only 5% of the course grade
- there are 50 (General Chemistry) to 70 (Organic Chemistry) questions per chapter
- the exam over this material will only contain exact questions from the homework. A three-chapter exam will contain 15-20 questions from the 150 (GChem) to 210 (Organic) question pool.
The Coach Approach incorporates several powerful features that significantly improve the learning experience:
- The importance of homework is reaffirmed as a process of learning complex associations that become stored in long-term memory.

- The class (players) along with the teacher (coach) and tutors/AI (support staff) work as a team so everyone can WIN WIN WIN.
-
- I'm not good at Chemistry.
- I studied the wrong things.
- If I studied 20 more hours, I would not have scored higher on the exam.
- Those questions came out of left field.
- The exam was too hard.
- The grading was too picky.
- I can't be successful.
- Effort is proportional to reward.
- Test anxiety is reduced when students have the exam question pool.
With the Coach Approach, students learn through daily success rather than failure. Basic knowledge is reinforced through Timed Repetitive Quizzes (TRQs), while homework builds the complex associations between concepts. As a result, students enter exams with confidence and demonstrate knowledge that has been stored in long-term memory.
More information and the amazing student results are found at The Coach Approach.
Chem21Labs Problem Types
Multiple Choice - contains 2 to 25 choices (text or image). Incorrect choices are greyed-out so students can't re-select them on subsequent tries.
Multiple Select - contains 2 to 20 choices (text or image). Students select 2 or more correct answers . . . . incorrect choices are greyed-out so students can't re-select them on subsequent tries. A sample question is shown to the right.
Which of the reactions can be classified as precipitation reactions?
Please select 2 correct answers from the following choices.
☐ Mg + HCl →
☐ NaHCO3 + HC2H3O2 →
☐ H2O + NH4Cl →
☐ H2O2 + KMnO4 →
☐ NaOH + HCl →
Short Answer and Fill-in-the-Blank - when an incorrect answer is entered, the program highlights the portions of the response that was entered correctly at the beginning and end. This provides immediate feedback, showing exactly where the mistake occurred. This type of problem is widely used for naming compounds in both General and Organic Chemistry. For example, if the correct answer is SO3 (Sulfur trioxide) and the student enters Sulfur dioxide, the program indicates which parts are correct (green) and which part is incorrect (red), allowing the student to identify and correct their error . . . . Sulfur ioxide .
Provide a name for SO3.
Sulfur
Your answer, Sulfur dioxide, is correct in the places shown: Sulfur ioxide
LewisDraw - students use a web-based drawing canvas to create the Lewis structure of a molecule, ion, radical, or resonance form. The tool provides structure-specific feedback, allows 3D rotation of the structure, and instructors can enable a mode where students must select the correct geometry. Click Open Lewis Draw to launch the application.
Chem21Draw -
Students complete organic chemistry homework by drawing answers on a web-based canvas. The application delivers highly detailed, structure-specific feedback because the grading system was developed specifically for this platform rather than relying on a SMILES library.
Chem21Draw automatically grades:
- Structures from names
- A single structure
- Multiple structures in labeled boxes. For example, a question such as "Draw the first four dicarboxylic acids" provides boxes labeled oxalic, malonic, succinic and glutaric acid. A correct structure placed in the wrong box is marked incorrect.
- Multiple structures for isomer or resonance questions, which may be placed anywhere on the canvas.
- Multistep synthesis
- Reagents are provided and students draw . . . .
- the product after each step. Each reaction arrow is followed by a box, and at least one atom of the correct answer must be inside the box to be counted correct.
- the final product.
- Students select reagents from a question-specific option pool and draw the organic product for each step.
- Reagents are provided and students draw . . . .
- Mechanisms can be drawn using one-electron and two-electron arrows. After an arrow is placed, its shape can be smoothly adjusted by curving it with Bézier control handles located at each end of the arrow.
Click Open Chem21Draw to launch the application.
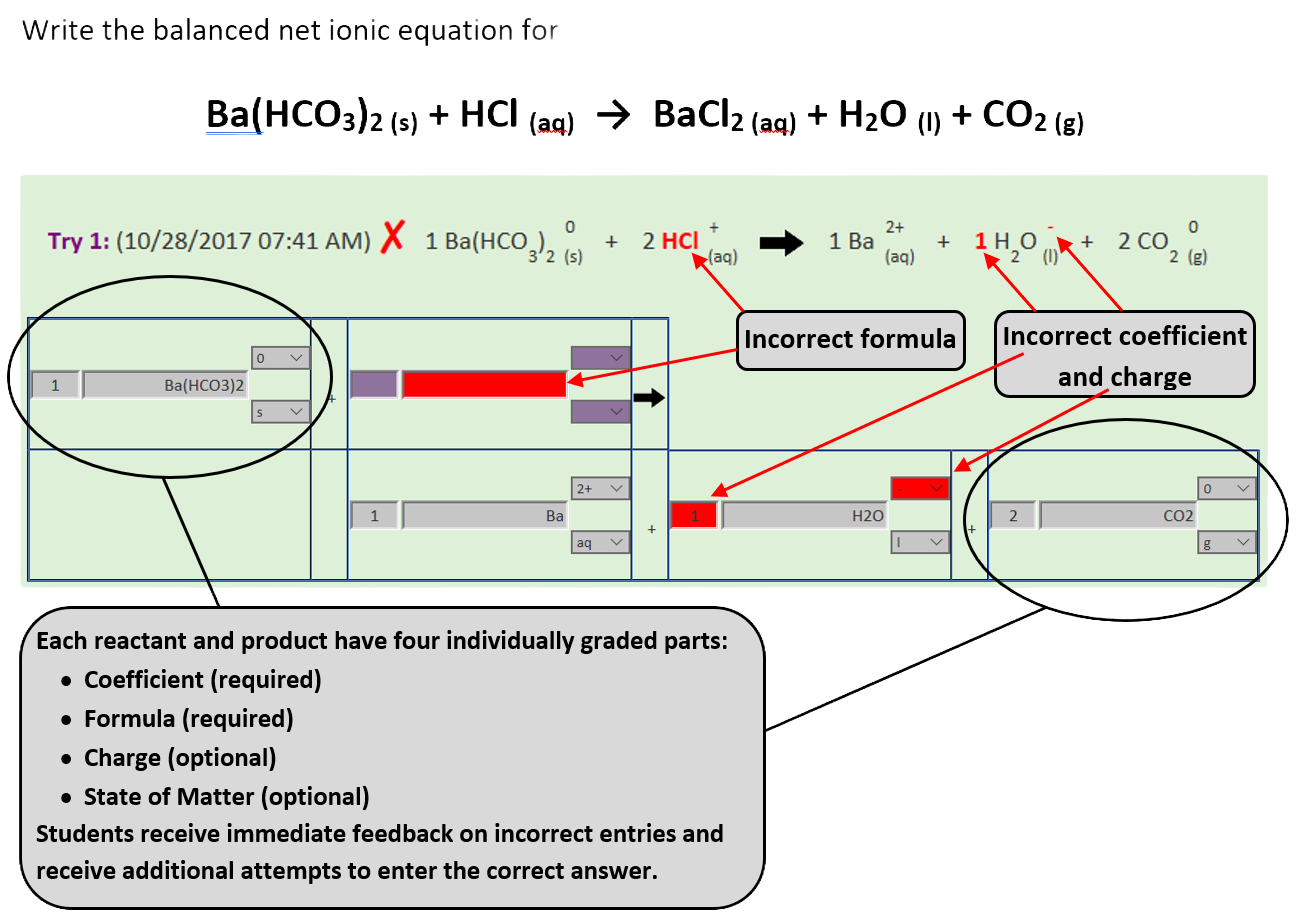
Balancing Chemical Equations - an equation template has been developed that contains 4 pieces of information for each chemical species
- two required - coefficient and chemical formula
- two optional - charge and physical state
Instructors can easily customize this template for different assignments. For example, one assignment may require students to enter only the coefficient, another may require the chemical formula and coefficient, a third may require a complete ionic equation (including charges and physical states), and a fourth may require the net ionic equation.
Balancing Nuclear Equations - a differently-arranged version of the equation grader is used to submit Nuclear Equations. The 4 pieces of information needed for a nuclear equation are coefficient, chemical symbol, atomic number, and mass number. Students get immediate feedback on their answer and have multiple attempts to earn credit.
Multiple Short Answers - this problem type is used for multi-part questions, where each part may require either a text or numeric response. A typical example is . . . .
Provide the following information for 56Fe0 where
56 is the mass and 0 is the charge.
Numeric Answers - a problem type in which the grading program checks whether a numeric answer falls within the tolerance range set by the instructor. The types of tolerances include:
- Absolute (±x)
- Percent (±x%)
- Significant Figure (±0-9 in the x SF)
- Decimal Place (±0 in the x DP)
Additionally, the question can include randomly generated numbers to give each student a unique answer. The instructor can also require responses in scientific notation and select either Chemical Rounding or Excel Rounding for the last two tolerance options.
For the following complex-thinking problems with numeric answers, Chem21 provides guided calculation tools that walk students step by step toward the correct solution.
- Dimensional Analysis - there are three interactive unit maps that build on each other.
- Map 1 - Length, Area, Mass and Volume
- Map 2 - Length cubed (Volume) is added along with density, amu, moles and molecules
- Map 3 - molarity is added along with a mole → mole conversion . . . . this effectively doubles Map 2.
- Empirical Formula Calculator
- pH Calculator - guides students step by step in calculating the pH of strong and weak acids and bases, including mixed solutions, buffers and diluted solutions.
Ordering - This problem type requires students to click and drag the displayed items into the correct order before submitting for grading. It is commonly used to sequence lab procedure steps as a pre-lab assignment (Open An Ordering Assignment). The instructor breaks the procedure into individual steps, and the student arranges them in the order they will be performed. It is also used in the Refractometer Tutorial to order the operation and reading steps.
The ordering problem type is also used fto arrange Periodic Table trends (electronegativity, ionization energy, atomic radius, metallic character, etc.) or types of electromagnetic radiation by their energy, wavelength, or frequency.
For example, students may be asked "Arrange the electromagnetic radiation in order of increasing wavelength." The image below illustrates the feedback students receive after their first submission.

